Abstract
The human microbiome is composed of trillions of micro-organisms living in symbiosis with the human body. The interest in the microbiome and the clinical impact of its disruption (dysbiosis) has grown exponentially in the past years, especially in the oncology field. Microbiota play an instructive role in chronic inflammatory disorders and determine response to checkpoint inhibitors cancers, such as melanoma. Increased inflammation in myeloproliferative neoplasms (MPN) drive many of the symptoms associated with this disease and inflammation also likely plays an important role as a driver of the disease. We hypothesized that the microbiome may be dysregulated in MPN which could contribute to the increased inflammatory cytokines seen in this disease and that the microbiome could also potentially impact symptom burden.
We compared the gut microbiota of 25 patients with classical MPN, essential thrombocythemia (ET), polycythemia vera (PV) and primary and secondary myelofibrosis (MF) to that of 25 normal controls. Whenever possible, co-inhabiting adults such as a spouse were used as normal controls. Exclusion criteria were acute illness and antibiotics treatment for three months preceding the study. Each participant collected three stool specimens in the course of one week and a subset of the group (n=20) had peripheral blood drawn. The stool microbiome was analyzed using 16S rRNA sequencing and plasma inflammatory cytokines (TNFα, IL-6, IL-8, IL-17a, IL-10, IFNγ, IFNa2, IL-22, IL1-β, GRO, IP-10) were measured using Luminex multiplex analysis. Participants also completed an intake survey which included symptom burden (MPN-SAF TSS) and dietary intake.
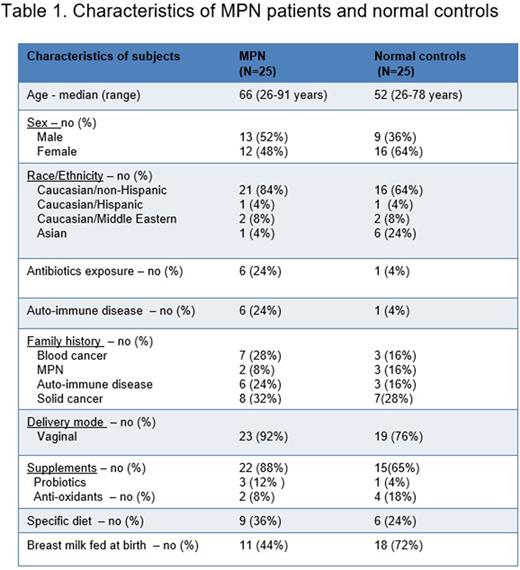
Characteristics of our MPN patient cohort and normal controls are shown in Table 1. Plasma levels of TNFα and IP-10 were significantly higher in the MPN group versus normal controls (respectively p=0.013 and p=0.0050). We divided the group into those with high (MPN-SAF > 20) and low (MPN-SAF ≤ 20) symptom burden, 7 (28%) patients had high symptom burden affecting considerably their quality of life. Fatigue and early satiety were the most common complaints. GRO (CXCL1) was significantly lower in MPN patients MPN patients with a lower symptom burden (p=0.0287) as compared to those with a high symptom burden.
The 16S rRNA gene sequencing did not reveal a significant difference between MPN patients and their healthy counterparts (PERMANOVA, p = 0.83), a large amount of variance in the microbiome data was explained by the individual (PERMANOVA, p= 0.0001), in concordance with other studies suggesting that the microbiome is highly individualistic. To determine whether certain taxa are differentially represented in MPN patients as compared to normal controls, a SIMPER test was used. Prevotellaceae, a family of bacteria implicated in chronic inflammatory conditions, was 20% higher in MPN patients than in normal controls.
To test which cytokines explained the most variance in the microbiome data, a distance-based linear model was used (DistLM). TNFα and IL-17a, taken alone, explain 18.7% and 14.5% respectively. The microbiome of patients with a high symptom burden was not significantly different from MPN patients with low symptom burden. We did find a significant difference between the microbiomes of patients treated with Ruxolitinib versus Hydroxyurea.
Taken together, this pilot study suggests that inflammation associated with MPN may be driving changes in the microbiome at a finer level, undetected when comparing broad microbial diversity metrics between health and disease. It is still not clear whether the inflammation first modulates the microbiome or whether the gut microbiota triggers the inflammatory signals driving the disease. A larger number of subjects may help answer these questions.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal